Re-purposing qXR for COVID-19
In March 2020, we re-purposed our chest X-ray AI tool, qXR, to detect signs of COVID-19. We validated it on a test set of 11479 CXRs with 515 PCR-confirmed COVID-19 positives. The algorithm performs at an AUC of
0.9 (95% CI : 0.88 - 0.92)on this test set. At our most common operating threshold for this version, sensitivity is0.912 (95% CI : 0.88 - 0.93)and specificity is0.775 (95% CI : 0.77 - 0.78). qXR for COVID-19 is used at over 28 sites across the world to triage suspected patients with COVID-19 and to monitor the progress of infection in patients admitted to hospital
The emergence of the COVID-19 pandemic has already caused a great deal of disruption around the world. Healthcare systems are overwhelmed as we speak, in the face of WHO guidance to ‘test, test, test’ [1]. Many countries are facing a severe shortage of Reverse Transcription Polymerase Chain Reaction (RT-PCR) tests. There has been a lot of debate around the role of radiology — both chest X-rays (CXRs) and chest CT scans — as an alternative or supplement to RT-PCR in triage and diagnosis. Opinions on the subject range from ‘Radiology is fundamental in this process’ [2] to ‘framing CT as pivotal for COVID-19 diagnosis is a distraction during a pandemic, and possibly dangerous’ [3].
Role of Radiography
The humble chest X-ray has emerged as the frontline screening and diagnostic tool for COVID-19 infection in a few countries and is used in conjunction with clinical history and key blood markers such as C-Reactive Protein (CRP) test and lymphopenia [4]. Ground glass opacities and consolidations which are peripheral and bilateral in nature are attributed to be the most common findings with respect to COVID related infections on CXRs and chest CTs. CXRs can help in identifying COVID-19 related infections and can be used as a triage tool in most cases. In fact, Italian and British hospitals are employing CXR as a first-line triage tool due to high RT-PCR turnaround times. A recent study [5] which examined CXRs of 64 patients found that in 9% of cases, initial RT-PCR was negative whereas CXRs showed abnormalities. All these cases subsequently tested positive for RT-PCR within 48 hours. The American college of Radiology recommends considering portable chest X-rays [6] to avoid bringing patients to radiography rooms. The Canadian Association of Radiologists suggest the use of mobile chest X-ray units for preliminary diagnosis of suspected cases [7] and to monitor critically ill patients, but have reported that no abnormalities are seen on CXRs in the initial stages of the infection.
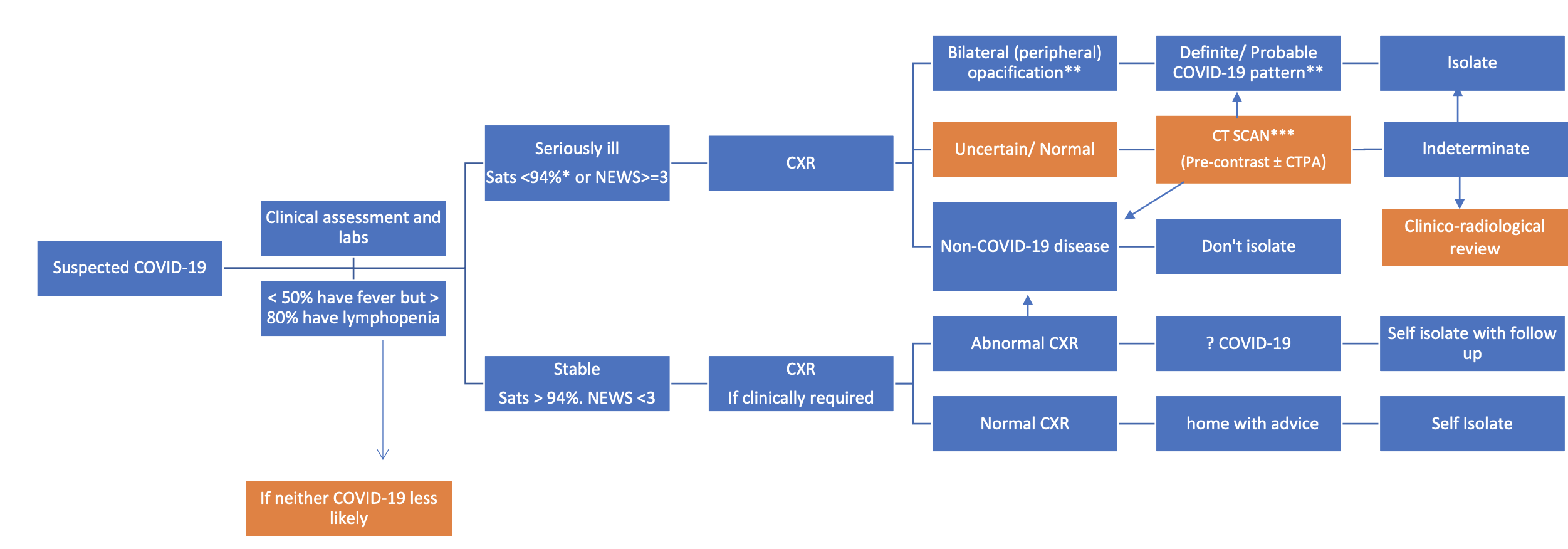
Radiology decision tool for suspected COVID-19 - The British Society of Thoracic Imaging [8]
As of today, despite calls for opening up imaging data on COVID-19 and outstanding efforts from physicians on the front-lines, there are limited X-ray or CT datasets in the public domain pertaining specifically to COVID. These datasets remain insufficient to train an AI model for COVID-19 triage or diagnosis but are potentially useful in evaluating the model – provided the model hasn’t been trained on the same data sources.
Building and evaluating qXR for COVID-19
Over the last month, customers, collaborators, healthcare providers, NGOs, state and national governments have reached out to us for help with COVID detection on chest X-rays and CTs.
In response, we have adapted our tried-and-tested chest X-ray AI tool, qXR to identify findings related to COVID-19 infections. qXR is trained using a dataset of 2.5 million chest X-rays (that included bacterial and viral pneumonia and many other chest X-ray findings) and is currently deployed in over 28 countries. qXR detects the following findings that are indicative of COVID-19: Opacities and Consolidation with bilateral and peripheral distribution and the following findings that are contra-indicative of COVID-19: hilar enlargement, discrete pulmonary nodule, calcification, cavity and pleural effusion.
These CE-marked capabilities have been leveraged for a COVID-19 triage product that is highly sensitive to COVID-19 related findings. This version of qXR gives out the likelihood of a CXR being positive for COVID-19, called Covid-19 Risk. Covid-19 Risk is computed using a post processing algorithm which combines the model outputs for the above mentioned findings. The algorithm is tuned on a set of 300 COVID-19 positives and 300 COVID-19 negatives collected from India and Europe.
Most new qXR users for COVID-19 are using it as a triage tool, often in settings with limited diagnostic resources. This version of qXR also localizes and quantifies the affected region. This capability is being used to monitor the progression of infection and to evaluate response to treatment in new clinical studies.
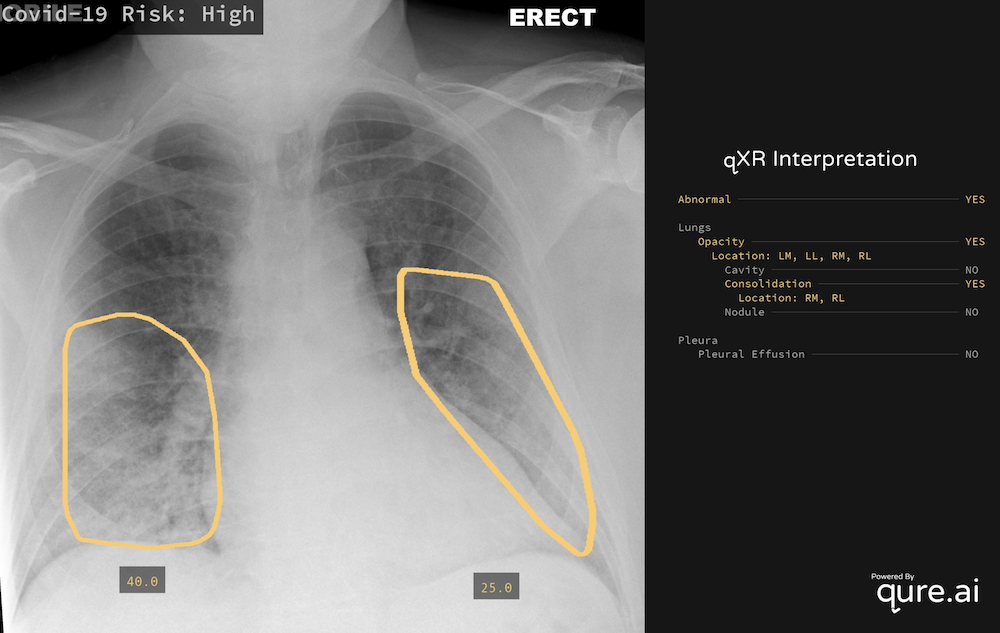
Sample Output of qXR [9]
Evaluation of the algorithm
We have created an independent testset of 11479 CXRs to evaluate our algorithm. The WHO [10] recommends a confirmatory diagnosis of COVID-19 using Reverse-Transcriptase Polymerase Chain Reaction (RT-PCR) - a specialised Nucleic Acid Amplification Test (NAAT) which looks for unique signatures using primers designed for the COVID-19 RNA sequence. Positives in this test set are defined as any CXR that is acquired while the patient has tested positive on RT-PCR test based on sputum/ lower respiratory and or upper respiratory aspirates/throat swab samples for COVID-19. Negatives in this test set are defined as any CXR which was acquired before the first case of COVID-19 was discovered.
The size of the negative set relative to the positive set was set to match the available prevalence in the literature [11]. The test set has 515 positives and 10964 negatives. Negatives are sampled from an independent set 250,000 CXRs. Negative set has 1609 cases of bilateral opacity and 547 cases of pulmonary consolidation in it (findings which are indicative of COVID-19 on a CXR), where the final diagnosis is not COVID-19. Negative set also has 355 non-opacity related abnormalities. This allowed us to evaluate algorithms ability to detect non COVID-19 opacities and findings, and is used to suggest alternative possible etiology and rule out COVID-19. We have used Area under Receiver Operating Characteristic Curve (AUC) along with Sensitivity and Specificity at the operating point to evaluate the performance of our algorithm.
| Characteristic | Value |
|---|---|
| Number of scans | 11479 |
| Positives | 515 |
| Negatives | 10964 |
| Normals | 9000 |
| Consolidation | 547 |
| Opacities | 1609 |
| Other Abnormalities | 355 |
Test set demographics
A subset (1000 cases) of this test set was independently reviewed by radiologists to create pixel level annotations to localize opacity and consolidation. Localization and progression monitoring capability of qXR is validated by computing the Jaccard Index between algorithm output and radiologist annotations.
Metrics
To detect signs of COVID-19, We have observed an AUC of 0.9 (95% CI: 0.88 - 0.92) on this test set. At the operating threshold, we have observed the sensitivity to be 0.912 (95% CI : 0.88 - 0.93) and specificity to be 0.775 (95% CI : 0.77 - 0.78). While there are no WHO guidelines yet for an imaging based triage tool for COVID, WHO recommends a minimum sensitivity and specificity of 0.9 and 0.7 for community screening tests for Tuberculosis [12], which is a deadly infectious disease in itself. We have observed a Jaccard index of 0.88 between qXR’s output and expert’s annotations.
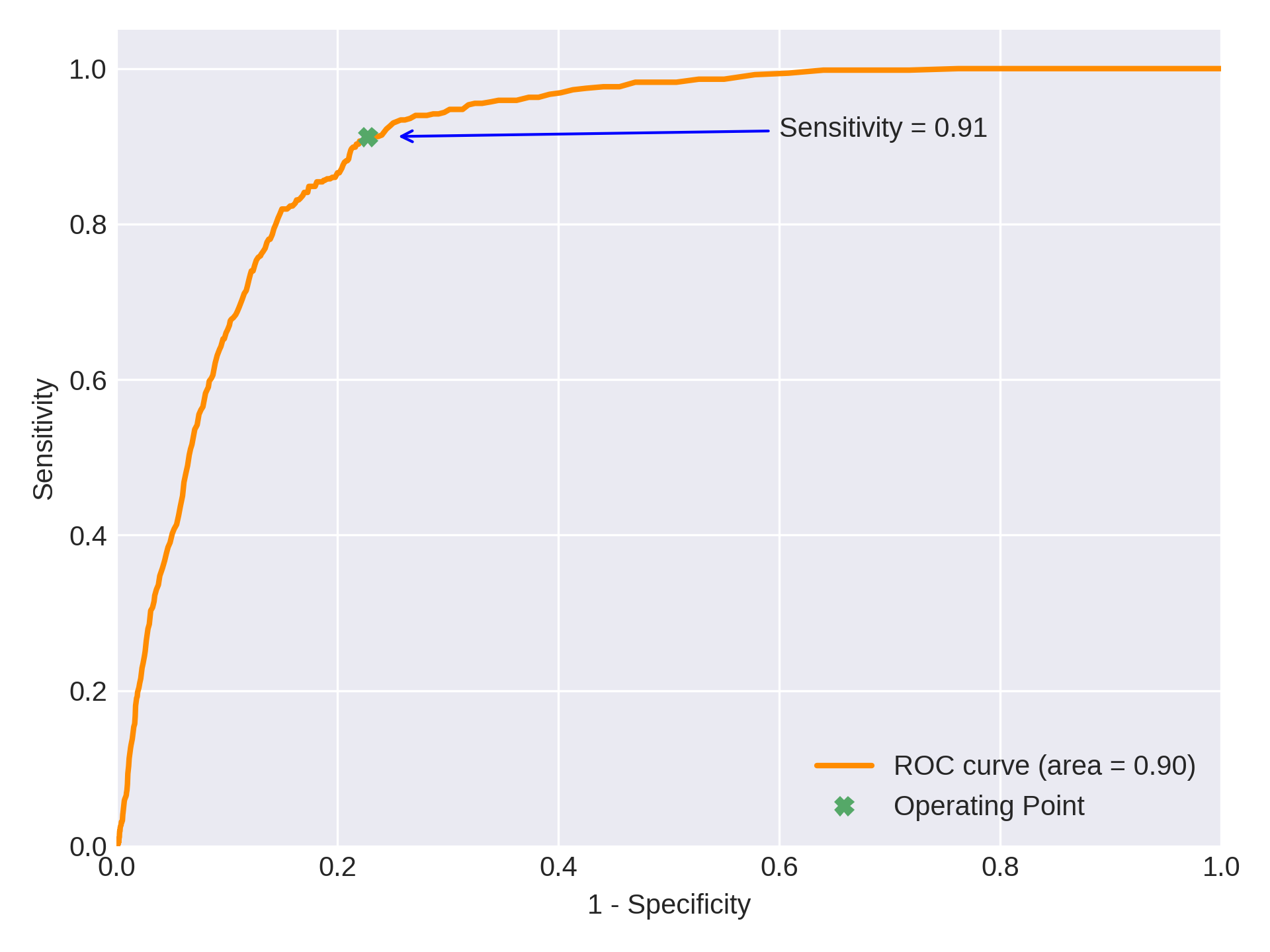
Receiver Operating Characteristic Curve
Deploying qXR for COVID-19
qXR is available as a web-api and can be deployed within minutes. Built using our learnings of deploying globally and remotely, it can interface with a variety of PACS and RIS systems, and is very intuitive to interpret. qXR can be used to triage suspect patients in resource constrained countries to make effective use of RT-PCR test kits. qXR is being used for screening and triage at multiple hospitals in India and Mexico.
San Raffaele Hospital in Milan, Italy has deployed qXR to monitor patients and to evaluate patient’s response to treatments. In Karachi, qXR powered mobile vans are being used at multiple sites to identify potential suspects early and thus reducing burden on the healthcare system.
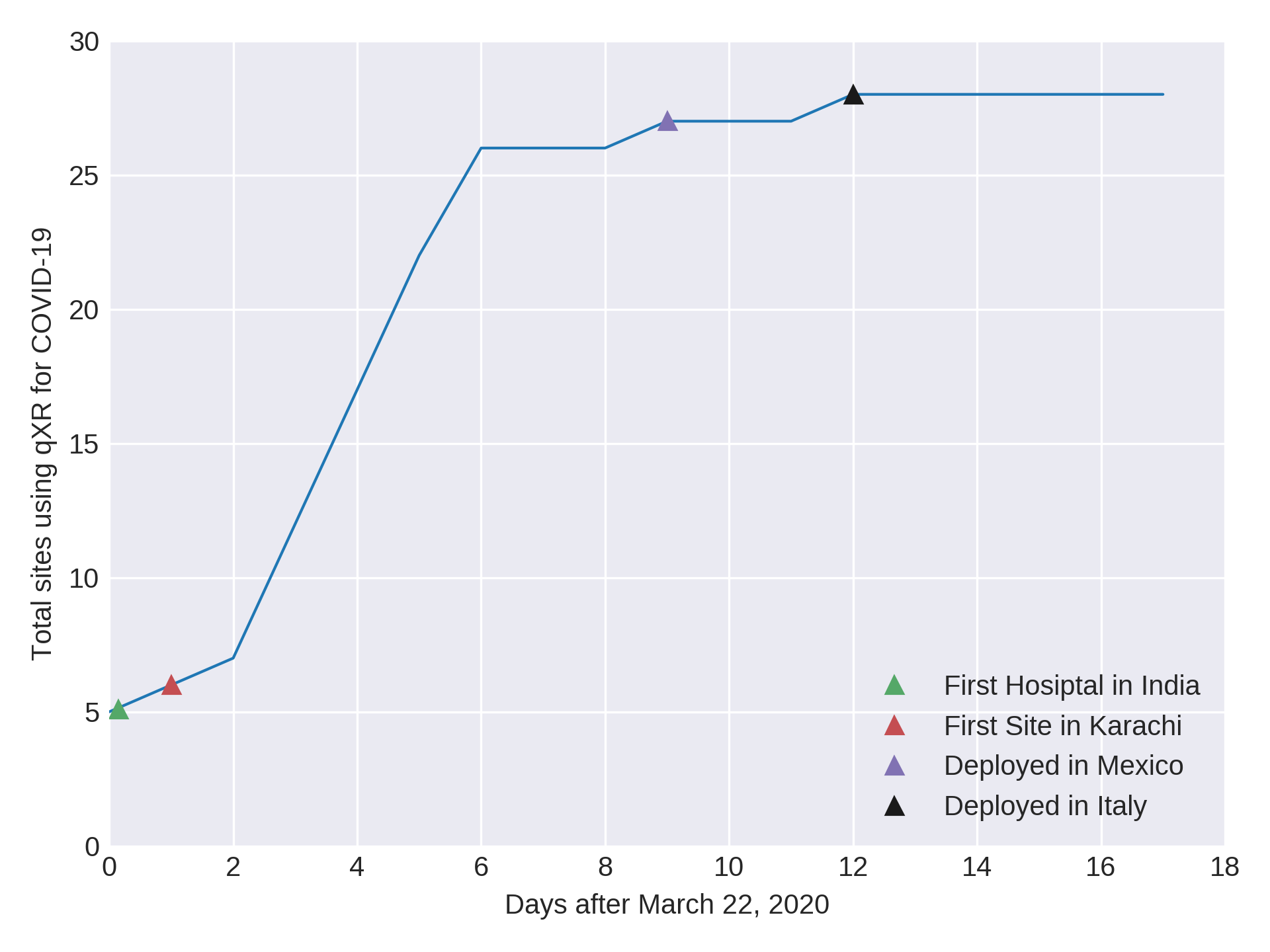
Timeline of qXR for COVID
In the UK, all the suspected COVID-19 patients presenting to the emergency department are undergoing blood tests and CXR [4]. This puts a tremendous amount of workload on already burdened radiologists as it becomes critical for radiologists to report the CXRs urgently. qXR, with its ability to handle huge workloads, provides significant value in such a scenario and thus reduce the burden on radiologists.
qXR can also be scaled for rapid and extensive population screening. Frontline clinicians are increasingly relying on chest X-rars to triage the sickest patients, while they await RT-PCR results. When there is high clinical suspicion for COVID-19 infection, the need for a patient with positive chest X-ray to get admitted in a hospital is conceivable. qXR can help solve this problem at scale.
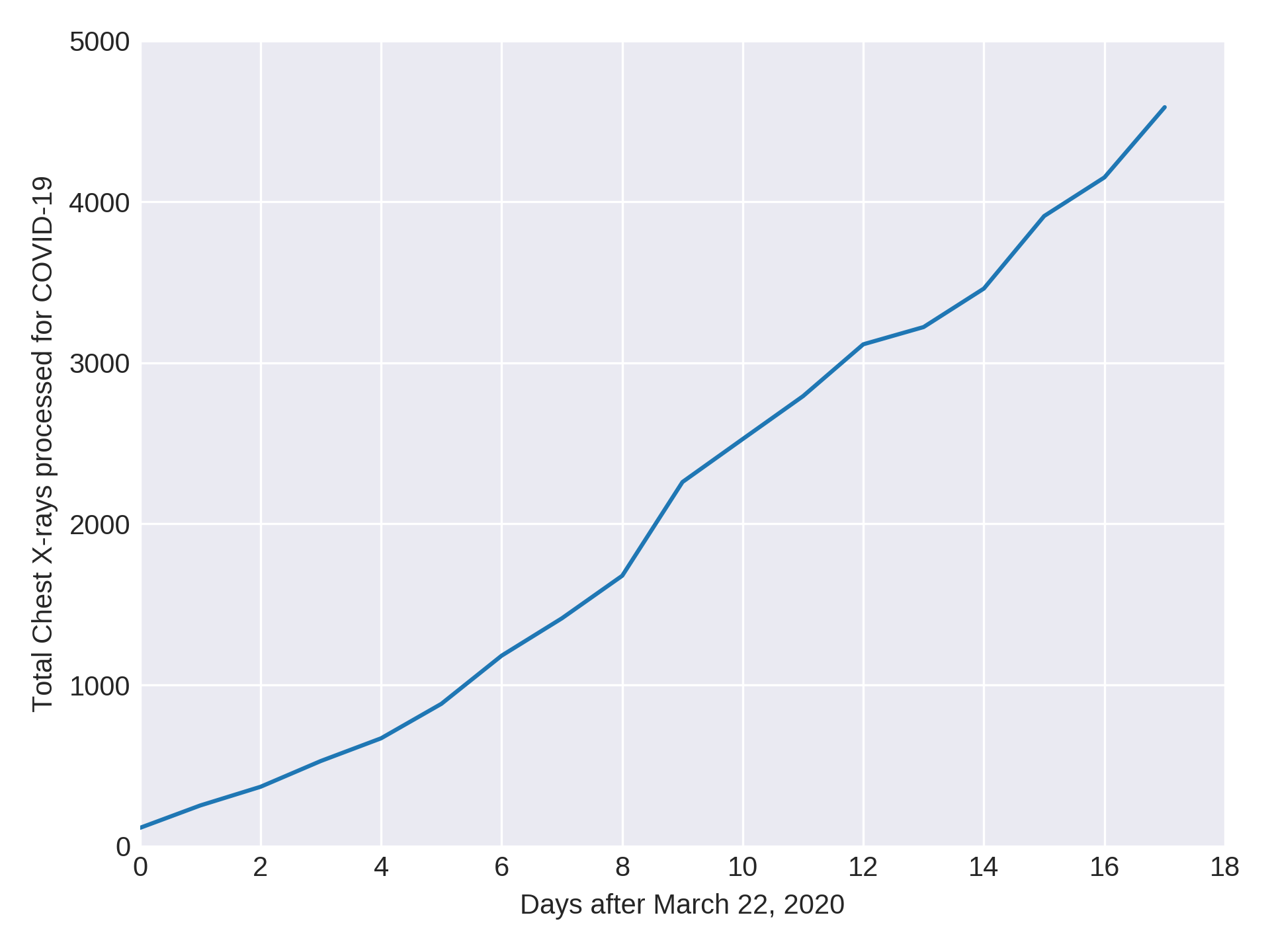
Impact of qXR for COVID-19
Work with us
With new evidence published every day, and evolving guidance and protocols adapting in suit for COVID-19, national responses globally remain fluid. Singapore, Taiwan and South Korea have shown that aggressive and proactive testing plays a crucial role in containing the spread of the disease. We believe qXR can play an important role in expanding screening in the community to help reduce the burden on healthcare systems. If you want to use qXR, please reach out to us.
References
- WHO Director-General’s opening remarks at the media briefing on COVID-19 - WHO, Accessed Apr 9, 2020.
- Imaging the coronavirus disease COVID-19 - Healthcare in Europe Website, Accessed Apr 9, 2020.
- Hope et al. A role for CT in COVID-19? What data really tell us so far - The Lancet, Mar 27, 2020
- Lessons from the frontline of the covid-19 outbreak - BMJ Blog, Accessed Apr 9, 2020.
- Wong et al. Frequency and Distribution of Chest Radiographic Findings in COVID-19 Positive Patients - Radiology, Mar 27, 2020.
- ACR Recommendations for the use of Chest Radiography and Computed Tomography (CT) for Suspected COVID-19 Infection - ACR, Accessed Apr 9, 2020.
- Lei et al. COVID-19 Infection: Early Lessons - Canadian Association of Radiologists Journal, Mar 12, 2020.
- Radiology decision tool for suspected COVID-19 - The British Society of Thoracic Imaging, Accessed Apr 9, 2020.
- Cohen et al. COVID-19 image data collection - arXiv:2003.11597, 2020
- Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases - WHO, Accessed Apr 9, 2020.
- Verity et al. Estimates of the severity of coronavirus disease 2019: a model-based analysis - The Lancet Infectious Diseases, Mar, 2020.
- High priority target product profiles for new tuberculosis diagnostics: report of a consensus meeting, tech. rep., World Health Organization, Apr 28-29, 2014.